- Blog
- Stasiun radio musik klasik jakarta
- Ns2 np3
- Smash bros 3ds challenges
- Xvideos andy sierra
- Tech utilities pc fix review
- Download autocad lt 2018
- Mag one a8 battery
- Fsx missions southwest
- Depriester chart with n propanol
- Emcopy vs robocopy large files network
- Malayalam film script format
- Far cry 4 ps4
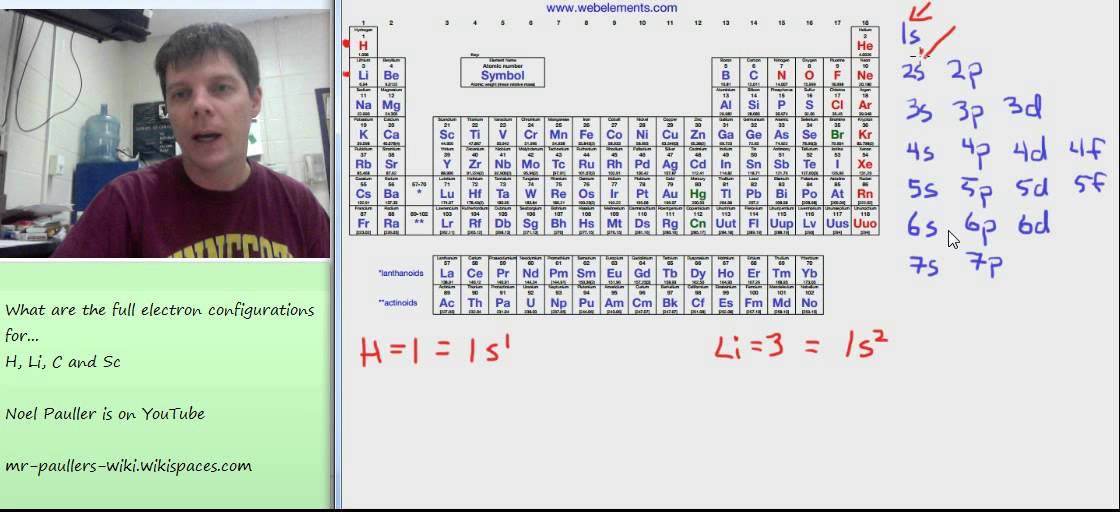
This can be removed as slag during reduction of Cuo to Cu. General electronic configuration ns2np5 is of halogen family. (A) ns2 np3 (B) n.s2 np4 (C) n.s2 np6 (D) ns2 np5 When copper pyrite is roasted in excess of air, a mixture of Cuo and Feo is formed.
General electronic configuration ns2np1 is of boron family. General electronic configuration ns2np2 is of carbon family. => outer electron configuration is ns, where n is the main energy level: 1, 2, 3, 4, 5, 6,7. General electronic configuration ns2np3 is of nitrogen family. What is the Trend in reactivity in the periodic table?ĭown the group from top to bottomThe reactivity of metals in the periodic table increases down the group due the increase in the number of energy levels which makes it easier for them to lose electron(s).Non-metals react by gaining electron(s), therefore, their reactivity decreases down the group because as the energy level increases the nuclear charge decreases.Īcross the period from left to right.Reactivity of metals decreases across the period because of an increase in nuclear charge which makes it more difficult for them to lose electron(s).On the other hand, reactivity of non-metals increases across the period due to increased nuclear charge that makes it easier for them to gain electron(s). Why are they the least reactive elements?Noble gases have an octet configuration, which means they have maximum number of electrons in their outermost energy levels.This makes them stable, which means they are inert.Examples of noble gases include neon, argon, krypton, etc. Noble gasesExplanation:Noble gases are elements in the last group of the periodic table. This increase in the atomic size is attributed to an increase in the number of shells.D. Nitrogen family: The elements of group 15 nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb) and bismuth (Bi), belong to configuration is ns2 np3. (iv) Atomic size: On moving down a group, the atomic size increases. As we move down a group, electronegativity decreases, owing to an increase in size. This is because of increasing atomic sizes. (iii) Ionization energy and electronegativity:įirst ionization decreases on moving down a group. Gp 13ns2 np1 Gp 14ns2 np2 Gp 15ns2 np3 Gp 16ns2 np4 Gp 17ns2 np5 Gp 18ns2 np6. This happens because of the inert pair effect. An element with the general electron configuration for its outermost electrons of ns2 np1 would be in which element group 2a 3a 4a 5a 8a. However, the stability of +5 oxidation state decreases down a group, whereas the stability of +3 oxidation state increases. Which of the following is the general electron configuration for the outermost electrons of elements in the alkaline earth group Ns1 Ns2 Ns2 np4 Ns2 np5 Ns2 np6 (n-1)d6.

In addition to the -3 state, N and P also show -1 and -2 oxidation states.Īll the elements present in this group show +3 and +5 oxidation states. The remaining elements of this group show a formal oxidation state of -3 in their covalent compounds. This can take place only with nitrogen as it is the smallest in size and the distance between the nucleus and the valence shell is relatively small. 1 ns1 10 ns2 (n 1) d8 2 ns2 11 ns1 (n 1) d10 3 ns2 (n 1) d1 12 ns2 (n. However, gaining electrons is very difficult as the nucleus will have to attract three more electrons. (ii) Oxidation states: All these elements have 5 valence electrons and require three more electrons to complete their octets. (ii) Oxidation states: All these elements have 5 valence electrons and require three more electrons to. The p-block aspects in the fourth duration and past will have actually thenoblegas core in addition to (n-1)d10. Group 17 elements have the general configuration of ns2 np5 F 2s2 2p5. Group 16 facets have the basic configuration the ns2 np4 O 2s2 2p4. Their general electronic configuration is ns2 np3. Their general electronic configuration is ns2 np3. Group 15 facets have the basic configuration the ns2 np3 N 2s2 2p3.

(i) Electronic configuration: All the elements in group 15 have 5 valence electrons.